2025 Boston Pharmaceutical Statistics Symposium
The Boston Pharmaceutical Statistics Symposium is the flagship event for the Boston Chapter. The Boston Pharmaceutical Statistics Symposium provides a unique venue for sharing statistical applications and research in the biotech-pharma industry and building connections among all colleagues of the Greater Boston area engaged in statistical practice. We welcome the participation of industry statisticians, academic researchers, university students, and any professionals interested in pharmaceutical statistical topics.
The Boston Chapter first organized the Boston Pharmaceutical Symposium in 2017. Since then, the symposium has become a very successful annual event. Because statistics is a fundamental element of this symposium, in 2024, the Boston Chapter and the symposium's scientific committee changed the name of the symposium from "Boston Pharmaceutical Symposium" to "Boston Pharmaceutical Statistics Symposium".
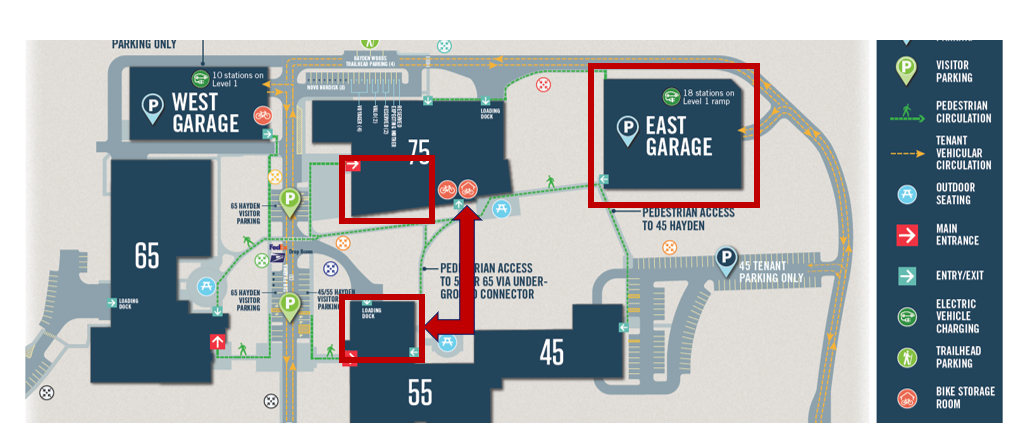
The 2025 Boston Pharmaceutical Statistics Symposium will be held at Novo Nordisk (75 Hayden Avenue), in Lexington, MA, on November 6 and November 7, 2025. On November 7, it is going to be a full-day event, featuring invited talks, a poster session, network opportunities, and much more. On November 6, the event will include two short courses.
Venue:
Novo Nordisk, 75 Hayden Avenue in Lexington
Information on the Bus route can be found here. The free parking location is the East Garage on the Novo Nordisk site.

Registration
Register Here for Symposium Main Event (Day 2)!
Register Here for Short Courses (Day 1, In-Person)!
Register Here for Short Courses (Day 1, Virtual)!
Update on Oct 31, 2025: last call for registration! Registration is now extended to Tuesday, Nov 4.
Registration Guide:
- All participants must register by October 31, 2025.
- Full-day Main Event: this event is for in-person participation only. Please use the first registration link if you would like to attend the symposium.
- Short Course (In-person): in-person is the primary format that is expected for the short course. If you wish to attend the short course in person, please use the second registration link.
- Short Course (Virtual-participation): the third registration link, labeled “Register Here for Short Courses (Day 1, Virtual)!” should only be used if you are planning to take the short course remotely.
- A valid photo ID is required and must be presented to the Novo Nordisk security on the event day. Please use the first name and last name that is shown on the photo ID for registration purposes. Email address will be used to contact registrants.
|
Thursday, November 6 – Short Course
|
Fees
|
|
morning short course - BCASA Member/Academia/Symposium SC Member
|
$108
|
|
morning short course - Non-BCASA Member
|
$135
|
|
morning short course - Student/Post-Doc
|
$50
|
|
afternoon short course - BCASA Member/Academia/Symposium SC Member
|
$108
|
|
afternoon short course - Non-BCASA Member
|
$135
|
|
afternoon short course - Student/Post-Doc
|
$50
|
|
2 short courses - BCASA Member/Academia/Symposium SC Member
|
$160
|
|
2 short courses - Non-BCASA Member
|
$200
|
|
2 short courses - Student/Post-Doc
|
$75
|
|
2 short courses - Student with Poster submission
|
$35
|
|
Short course virtual attendance
|
Same fees as in-person short course attendees
|
|
Friday, November 7– Full-day Main Event
|
|
|
Student
|
$35
|
|
BCASA Member/Post-Doc/Symposium SC Member
|
$85
|
|
Non-BCASA Post-Doc, with 1-year membership
|
$95
|
|
Non-BCASA Member, with 1-year membership
|
$125
|
|
Non-BCASA Member
|
$115
|
Agenda
Thursday, November 6, 2025
|
8:55 am - 9:00 am
|
Welcome: Gautier Paux
|
| 9:00 am - 12:00 pm: Short Course |
| Susan Gruber |
TMLE in Drug Development |
| Moderator: Maria Kudela |
| Lunch Break |
| 1:00 pm - 4:00 pm: Short Course |
| Haolin (Leo) Li |
An Overview of Machine Learning Methods for Survival Data |
| Moderator: Ting Wang |
Friday, November 7, 2025
Speakers Bios
Short Courses:
Haolin (Leo) Li, PhD
Haolin (Leo) Li, PhD is an Assistant Professor of Biostatistics at the Boston University School of Public Health. His research areas include survival analysis, machine learning, clinical trials, study design, and data synthesis. Dr. Li’s work focuses on developing and applying advanced statistical and machine learning methods in studies with outcome-dependent sampling designs. He also conducts research in data synthesis and subgroup analysis in clinical trials, biomarker-integrated clinical trial design, and statistical methods for complex survey data. He is also actively involved in interdisciplinary collaborations, particularly in epidemiological and biomedical research.

Susan Gruber, PhD
Susan Gruber, co-founder of software start-up TL Revolution, is a biostatistician and computer scientist who is working to providing accessible, expert-driven tools for targeted learning. She previously founded Putnam Data Sciences, a statistical consulting firm specializing in causal inference and predictive modeling to support public health and regulatory decision making. In addition to leading and collaborating on FDA-funded projects to improve evaluation of drug safety and effectiveness, Dr. Gruber developed the first open source R package for TMLE and has an extensive record of publications, presentations, and workshops on Targeted Learning.

Keynote Speakers:
Brian Millen, PhD
Brian A. Millen, Ph.D., is Vice President, Quantitative Sciences at Biogen, where he provides strategic and administrative leadership to the Biostatistics, Real World Data Analytics, and Epidemiology organizations. In this role, he ensures analytics-driven decision making in support of Biogen’s research and development portfolio. Prior to joining Biogen, Dr. Millen held multiple technical and senior leadership roles within the statistics organization Eli Lilly and Company. His career in biopharmaceutical statistics spans more than two decades, and he is known for establishing team cultures of high performance, inclusion and innovation.
Dr. Millen’s service to the profession includes launching and leading development programs for students and early career professionals, teaching statistics to non-statistics colleagues through various trade organizations, multiple leadership and advisory roles within the American Statistical Association (ASA) and the International Biometrics Society, chairing/co-chairing numerous workshops or conferences serving statisticians at various career levels, and publishing statistical methods and applications.
Dr. Millen holds a Ph.D. in Statistics from The Ohio State University and a BA in Mathematics from the University of Georgia. He is a fellow of the American Statistical Association, and he was recently elected the 122nd President of the ASA.

Josh Chen, PhD
Dr. Josh Chen is the Vice President of Biostatistics at Vertex Pharmaceuticals. Josh and his team provide biostatistical leadership in developing transformative medicines for people with serious and genetic diseases. Prior to Vertex, Dr. Chen held leadership positions at Sanofi Pasteur, the vaccines company of Sanofi, and at Merck Research Laboratories. His main research interest is about clinical trial designs, including group sequential design, adaptive designs and multiregional clinical trials. In his 25-year career in developing medicines, Josh has authored/co-authored two books on best practices for simultaneous global development, and 50+ papers on statistical methods and clinical trial applications in peer-reviewed journals. Dr. Chen received his PhD in Statistics from the University of Wisconsin-Madison and Bachelor in Probability and Statistics from Peking University. He is a Fellow of the American Statistical Association.

Invited Talk Sessions:
Kentaro Takeda, PhD
Dr. Kentaro Takeda is Senior Director, Head of Innovative Statistics at Quantitative Sciences and Evidence Generation at Astellas Pharma Global Development, Inc. His research interests cover Bayesian clinical trial designs, including dose-finding trials, basket trials, efficacy and safety monitoring, use of real-world data, and oncology statistics. Dr. Takeda has published more than 50 peer-reviewed papers in this research area. Dr. Takeda leads an innovative statistics group at Astellas and has implemented novel statistical approaches, including his proposed methods, in new drug development. Dr. Takeda also serves as an associate editor of several academic journals.

Yoni Sidi, PhD
Jonathan (Yoni) Sidi is a Director of Clinical Modeling at Sanofi with 15 years of experience modeling clinical data across CNS, immunology, oncology, and cardiovascular therapeutic areas. As an active R community developer for the past decade, Yoni contributes to the cross-industry openstatsware team developing R solutions for MMRM and Bayesian MMRM models. He's passionate about creating collaborative tools for model development and stakeholder engagement to drive clinical decision-making.

Junwei Lu, PhD
Junwei Lu is an Assistant Professor in the Department of Biostatistics at Harvard School of Public Health. His work focuses on the intersection of statistical artificial intelligence and translational clinical research by leveraging electronic health records and medical imaging data.

Krishna Padmanabhan, PhD
Krishna Padmanabhan is a biostatistician with 20+ of experience in the pharmaceutical industry, including leadership roles at Pfizer, Cytel and (currently at) Madrigal Pharmaceuticals. His statistical expertise spans multiple therapeutic areas, with a focus on Rare Diseases, Oncology, CVMET and Neuroscience. He has contributed to numerous regulatory submissions and market access dossiers and has helped design several innovative clinical trials including Bayesian and adaptive methods. Krishna has also held Pharma roles in commercial development, analytics, and market access in the past, and currently serves as adjunct faculty member at the University of Pennsylvania, where he teaches Probability, Statistics, and Machine Learning.

Jake Gagnon, PhD
Dr. Jacob Gagnon is a director of biostatistics at Biogen and leads a team of medical researchers in the areas of neurology and immunology. He leads statistical methodology development efforts for the latest omics technologies (ie spatial transcriptomics, scRNAseq, single cell proteomics, etc), performs preclinical research, is a core member of the text mining center of excellence, and leads a ML/DL focus group. His team’s research interests include deep learning, machine learning, translational biology, omics analysis, computer vision, and text mining. He obtained a PhD in statistics from UMASS Amherst and did postdoctoral work in biostatistics at WPI. After his postdoctoral work, he did biostatistics research for Abbvie, Roche, and then Biogen. He has authored/co-authored around 20 publications including three in Nature journals. Additionally, he has won multiple awards including: a winner of the PHUSE/FDA innovation challenge, NESDI’s best application of theory award, and Wiley’s highly viewed article award.

Foroogh Shamsi, PhD
Foroogh Shamsi, PhD is a Senior Data Scientist in the AI/ML Sciences team at Novo Nordisk. Her work focuses on advancing the application of machine learning, statistical methods, and targeted learning and causal inference in clinical research to improve randomized clinical trials. She collaborates on initiatives to implement modern covariate adjustment methods and contribute to the development of R packages designed to simulate and optimize clinical trial designs.

Yunqi Zhao, PhD
Yunqi Zhao is a Senior Manager, Statistics, in the Statistical and Quantitative Sciences (SQS) department at Takeda Pharmaceutical Company. She serves as a study lead statistician for oncology clinical trials. Her research interests include dose optimization, seamless phase I/II study design, machine learning, and historical data borrowing using Bayesian techniques to support decision-making and enhance clinical trial development. Within Takeda, Yunqi has led and contributed to internal working groups aimed at fostering innovations. She is passionate about bringing innovations to clinical development.

Sponsorship
We would like to warmly invite your organization to join us as a sponsor of the 2025 symposium. Sponsorship offers a unique opportunity to connect with a highly engaged audience of statisticians and data scientists while supporting the professional growth and collaboration within our industry. More details can be found in the sponsorship letter.
Venue Sponsor:

Platinum Sponsors:

Gold Sponsors:


Silver Sponsors:

Poster Sponsor:

Short Course Sponsor:

2025 Boston Pharmaceutical Statistics Symposium Scientific Committee
Symposium Co-Chair and Program Chair of the Boston Chapter of ASA
Gautier Paux, Sanofi
Symposium Co-Chair and Chair of Speakers and Short Course Subcommittee
Maria Kudela, Pfizer
Symposium Vice-Chair and Venue Host
Tu Xu, Novo Nordisk
Symposium Vice-Chair, Chair of Communication Subcommittee, and Webmaster of the Boston Chapter of ASA
Kush Kapur, argenx, Inc.
Symposium Past-Chair and In-charge of student volunteers, Boston local liaison
Kristin Baltrusaitis, Center for Biostatistics in AIDS Research, Harvard T.H. Chan School of Public Health
Symposium Past Co-Chair and Past President of the Boston Chapter of ASA
Wenting Cheng, Biogen
Symposium Past Co-Chair
Zhaoyang Teng, Astellas
President of the Boston Chapter of ASA
Jianchang Lin, Takeda
Symposium Scientific Committee Members
Hailu Chen, Alkermes
Brooks Clark, Cytel
Jinghui Dong, Kite, Gilead Science
Jimmy Efird, VA CSPCC - Boston
Kosalaram Goteti, Seaport Therapeutics, Inc.
Ina Jazic, Vertex
Fotios Kokkotos, Boston University
Haolin Li, Boston University
Xihao Li, UNC-Chapel Hill
Rachel Liu, Takeda
Jameson Luks, MaxisIT
Abigail Sloan, Pfizer
Ting Wang, Biogen
Disa Yu, Sanofi
Jing Yu, Novo Nordisk
Abstracts for Poster/Lightning Talk
Participants interested in sharing their work at the symposium are encouraged to submit an abstract to the poster session. Presentations may be given on any topic related to pharmaceutical or academic statistics. Any topics related to transforming clinical development through data science, innovative design, statistical excellence, and other broad topics are encouraged. Some additional highlights:
● Selected abstracts will be invited for a lightning talk before the poster session
● A student poster award competition will be hosted
Opens: July 21, 2025
Closes: September 17, 2025
To submit an abstract, you will need the following:
1. Authors’ names
2. Presenting author’s email and affiliation(s)
3. Abstract title
4. Abstract text (250-word limit)
5. Abstract keywords (max 6)
SUBMISSION CLOSED
All presenters are expected to register for the symposium and are responsible for printing and transporting the poster. For additional information, please contact: ASA.Boston.Pharm.Symposium@gmail.com.